|
ProductImages/106036/big/79g-04317.png' alt='Microsoft Office 2010 Cost' title='Microsoft Office 2010 Cost' />Im coming to the end of my trial period with Microsoft Office 2013.Over the weeks it has been a reasonably solid experience with one or two quirks causing me no.Buy the Office thats best for youOffice 365 gives you the power and flexibility to get things done from virtually anywhere.The much awaited Office 20162010 full version is now available for PC users.One can get them from here.Microsoft office 2.Ms office 2.Free Crack Corner.Microsoft include many new features in this version.Recently, Microsoft release Office 2.Just Follow These Simple Steps 1.Disconnect internet.Start Word Or any other Office Document 3.If the activation window appears close it.Go to menu File, Help, Change Product Code, and then enter key.Serial Keys 8.CH G4. FPY HMH 4.R 3.GCK 6. D9. BH 3.W3.MQ H7.
0 Comments
Archives/edgar/data/1035002/000119312514065039/g680806680806z0032.gif' alt='Naphtha Cracking Reaction Chemistry' title='Naphtha Cracking Reaction Chemistry' />The American Petroleum Institute API is the only national trade association that represents all aspects of Americas oil and natural gas industry.Our more than.Understanding Naphtha and Ethane Cracking Processes By Frank Caprio On June 6, 2017.Ethylene because its molecules have very distinctive and useful chemical.Organic Chemistry GCE Study Buddy 1.Ethyl ethanoate is made by esterification.The table below gives the relative molecular masses for the reactants and products in the reaction.Mr ethanoic acid 6.In an experiment, 3.What was the percentage yield of ethyl ethanoate Propanoic acid has the following structural formula.Draw the structural formula of the ester formed when propanoic acid reacts with ethanol.Solution.Ethanol, C2. H5. OH is used in Brazil as a liquid fuel instead of gasoline petrol.The ethanol is manufactured by a fermentation process from sugar cane.How would you test for this gasWhen ethanol is completely burnt in air, it forms carbon dioxide and water.One mole of ethanol will release 1.J of heat energy during this reaction.Construct the balanced equation for the burning of ethanol.The mass of 1 litre of ethanol is 7.How much heat energy will be released when 1 litre 1dm.When completely burnt, 1 litre of gasoline releases 3.J of heat energy.Comparing ethanol and gasoline as liquid fuels, suggesti.Solution.A mixture of sugar cane which contains glucose with some yeast is allowed to ferment in a closed container in the absence of air for some time.Difference-Between-Combustion-and-Oxidation-Reactions-of-Ethanol-2.jpg' alt='Naphtha Cracking Reactions' title='Naphtha Cracking Reactions' />Naphtha Cracking Reaction Of AlkanesModern steam cracking plants are very large, usually producing 12 million tonnes of products annually and several have been built recently that can have an output of.Fluid catalytic cracking FCC is one of the most important conversion processes used in petroleum refineries.It is widely used to convert the highboiling, high.C6.H1. 2O6 yeast 2.C2. Baixar Livros Gratis Gospel Em Pdf Files . H5. OH 2. CO2.This gas can be tested by passing it through limewater and the solution turns milky white precipitate formed2bi.C2.H5. OH l 3. O2 g 2.CO2 g 3.H2. O l2bii.Mr of ethanol 2 x 1.J2ci.Burning ethanol produces less carbon dioxide, which is one of the components responsible for the Greenhouse effect.Burning gasoline produces more energy.The following questions refer to compounds with the structures drawn below.Using the letters A to F as appropriate, answer the following.Which compound is an unsaturated hydrocarbon Which compounds are members of the same homologous seriesWhich compound reacts with sodium carbonate Which compounds are isomers of each other Solution.E3b.B, F3c. C3d.A, D4.Olive oil contains oleic acid which is unsaturated.By naming the reagent and giving the observation, describe a simple test to confirm that olive oil contains an acid.Explain what is meant by unsaturatedaiii.By naming the reagent and giving the observation, describe a simple test to confirm that oleic acid is unsaturated.The molecular formula of oleic acid is C1.H3.CO2. H. How many double bonds between carbon atoms are present in one molecule of oleic acidExplain your answer.Solution.Gas turns limewater milky.An unsaturated compound is one that contains multiple bonds eg double bonds.Explain If oleic acid does not contain CC double bond, its formula should be.C1.H3. 5CO2. H Cn.H2n1.CO2. H. Since its formula has 4 hydrogen less than 3.The structure of fluorochloroethane is shown belowa.Suggest a name for, and sketch the structure of, the macromolecule formed when fluorochloroethene polymerizes.When 2.Calculate the percentage yield.Name and give a use for a commercially available polymer containing a halogen.Solution.Block Usb Port Windows Xp Registry Editor .Theoretically, 2.Therefore yield 1.PVC.It is used as plastics.Ethyne, C2.H2, burns in oxygen to give a very hot flame.The temperature is so high that the flame can be used to cut metal.Name the two products of the complete combustion of ethyne.Describe a chemical test for one of these productsb.Calcium carbide, Ca.C2, is a greyish white solid.Ethyne can be made by the reaction of calcium carbide with water.Calcium hydroxide, CaOH2 is the other product of the reaction.Construct an equation, including state symbols, for the reaction between carlcium carbide and water.The mixture obtained after this reaction was tested using Universal Indicator.What was the final color of the Universal Indicator Explain your answer.Solution.Bubble the gas through limewater.A white precipitate would be observed.Ca.C2 s 2. H2. O l CaOH2 aq C2.H2 g6bii.Blue. CaOH2 is alkaline.The following terms are used in the description of organic compounds.From this list, choose two terms which can be applied to each of the compounds below.One of the general characteristics of homologous series is that all the members can be represented by a general formula.What is the general formula of the alkene series Give one other general characteristics of homologous series.Solution.Cn. H2n. 7bii. The members show similar chemical reactions.Heptane is an alkane with seven carbon atoms per molecule.It is a colorless liquid with a boiling point of 1.C.Predict the results of the tests for both heptane and water.Fructose is a carbohydrate.One form of fructose has the following structure.Solution.C7.H1. 68aii. test 1 add anhydrous copperII sulphateheptane solid copperII sulphate remains white and insolublewater solid turns blue and then dissolves to give a blue solutiontest 2 ignite with a flameheptane burns with a luminous flamewater the flame dies off.C6.H1. 2O6. 8bii.The molecular formula seems to suggest 6 units of carbon, and hydrogen and oxygen in the ratio 2 1 just like water C6H2.Hydrogen can be manufactured by the reaction between methane and steam.Name a source of the methane required for this reaction.Give the equation for this reaction between methane and steam.Calculate the maximum volume of hydrogen, measured at r.Margarine is manufactured using the addition reaction between hydrogen and a vegetable oil.The diagram represents the structure of a common plastic.Name this plastic and draw the structure of the monomer from which it is made.This plastic is non biodegradable.Explain the meaning of this term and describe the problems which this property creates.If this plastic is burned, a thick, black smoke and a very acidic gas are produced.Solution.CH4 g H2. O g CO g 3.H2 g9aiii.Mr of CH4 1.CH4 1.H2 formed 3 molvolume of 1 mole of gas at r.H2 formed 3 x 2.Nickel catalyst, high pressure.PVC9cii.Non biodegradable means cannot be broken down by biochemical reactions by bacteria.Ethene is an unsaturated hydrocarbon.Ethene is made by the cracking of long chain hydrocarbon molecules such as C1.H3.Construct an equation to illustrate the cracking of C1.H3.Ethene is bubbled through aqueous bromine to form 1,2 dibromoethane.What would you observe during the reactionGive the name of the product and the conditions needed for the reaction of ethene with water.Calculate the volume of carbon dioxide, measured at r.Ethene can be made into polyethene.Draw the structure of polyethene.Solution.An unsaturated compound is one that contains multiple bonds eg CC1.C1.H3. 4 3. C2.H4 C1.H2. Decolorization of brown bromine.C2.H4 g 3. O2 g 2.CO2 g 2.H2. O lMr of C2.H4 2.C2.H4 give 4. 8 dm.CO2 at r.C2. H4 give 1.CO2 at r. t. p. 1.The following is a list of formulae of organic compounds.CH3.COOHC5. H1. 2C2.H5.OHC3. H6. C7.H1. 4Which of the above formula fit the following descriptions VI to give ethanoic acid.Solution.CH3.COOH1. 1b. CH3.COOH or C2.H5. OH1.C3. H6 or.C7. H1.CH3.COOH and C2. H5.OH1.C3. H6 or C7.H1.C2. H5. OH1. 2.Ethanol, CH3.CH2.OH is a liquid fuel.Ethanol can be manufactured either from glucose C6.H1.O6 or from ethene.Briefly describe the manufacture of ethanol from glucose.Include the balanced equation in your answer.Draw the displayed formula for ethenebii.Name the substance that reacts with ethene to make ethanolbiii.Give the conditions needed for this reaction.Solution.Yeast cells are added to a solution of glucose.The mixture is kept at 1.C under anaerobic conditions.Glucose will be broken down by zymase enzyme in yeast cells to form ethanol and carbon dioxide.Ethanol is obtained by fractional distillation of the mixture.C6.H1. 2O6 2. C2.H5.OH CO2. C, 7.Styrene butadiene rubber is a synthetic rubber.It is made by polymerising a mixture of the monomers butadiene and styrene.What type of polymerization will take place when the monomers polymeriseCracking chemistry Wikipedia.In petrochemistry, petroleum geology and organic chemistry, cracking is the process whereby complex organicmolecules such as kerogens or long chain hydrocarbons are broken down into simpler molecules such as light hydrocarbons, by the breaking of carbon carbon bonds in the precursors.The rate of cracking and the end products are strongly dependent on the temperature and presence of catalysts.Cracking is the breakdown of a large alkane into smaller, more useful alkanes and alkenes.Simply put, hydrocarbon cracking is the process of breaking a long chain of hydrocarbons into short ones.This process might require high temperatures and high pressure.More loosely, outside the field of petroleum chemistry, the term cracking is used to describe any type of splitting of molecules under the influence of heat, catalysts and solvents, such as in processes of destructive distillation or pyrolysis.Fluid catalytic cracking produces a high yield of petrol and LPG, while hydrocracking is a major source of jet fuel, Diesel fuel, naphtha, and again yields LPG.History and patentseditAmong several variants of thermal cracking methods variously known as the Shukhov cracking process, Burton cracking process, Burton Humphreys cracking process, and Dubbs cracking process Vladimir Shukhov, a Russian engineer, invented and patented the first in 1.Russian Empire, patent no.November 7, 1.One installation was used to a limited extent in Russia, but development was not followed up.In the first decade of the 2.American engineers William Merriam Burton and Robert E.Humphreys independently developed and patented a similar process as U.S.June 8, 1. 90. 8.Among its advantages was the fact that both the condenser and the boiler were continuously kept under pressure.In its earlier versions however, it was a batch process, rather than continuous, and many patents were to follow in the USA and Europe, though not all were practical.In 1.American Sinclair Oil Corporation visited Shukhov.Sinclair Oil apparently wished to suggest that the patent of Burton and Humphreys, in use by Standard Oil, was derived from Shukhovs patent for oil cracking, as described in the Russian patent.If that could be established, it could strengthen the hand of rival American companies wishing to invalidate the Burton Humphreys patent.In the event Shukhov satisfied the Americans that in principle Burtons method closely resembled his 1.Russian oil industry could easily build a cracking apparatus according to any of the described systems without being accused by the Americans of borrowing for free.At that time, just a few years after the Russian Revolution, Russia was desperate to develop industry and earn foreign exchange, so their oil industry eventually did obtain much of their technology from foreign companies, largely American.At about that time however, fluid catalytic cracking was being explored and developed and soon replaced most of the purely thermal cracking processes in the fossil fuel processing industry.The replacement was however not complete many types of cracking, including pure thermal cracking, still are in use, depending on the nature of the feedstock and the products required to satisfy market demands.Thermal cracking remains important however, for example in producing naphtha, gas oil, and coke, and more sophisticated forms of thermal cracking have been developed for various purposes.These include visbreaking, steam cracking, and coking.ChemistryeditA large number of chemical reactions take place during the cracking process, most of them based on free radicals.Computer simulations aimed at modeling what takes place during steam cracking have included hundreds or even thousands of reactions in their models.The main reactions that take place include InitiationeditIn these reactions a single molecule breaks apart into two free radicals.Only a small fraction of the feed molecules actually undergo initiation, but these reactions are necessary to produce the free radicals that drive the rest of the reactions.In steam cracking, initiation usually involves breaking a chemical bond between two carbon atoms, rather than the bond between a carbon and a hydrogen atom.CH3.CH3 2 CH3Hydrogen abstractioneditIn these reactions a free radical removes a hydrogen atom from another molecule, turning the second molecule into a free radical.CH3 CH3.CH3 CH4 CH3.CH2Radical decompositioneditIn these reactions a free radical breaks apart into two molecules, one an alkene, the other a free radical.This is the process that results in alkene products.CH3.CH2 CH2CH2 HRadical additioneditIn these reactions, the reverse of radical decomposition reactions, a radical reacts with an alkene to form a single, larger free radical.These processes are involved in forming the aromatic products that result when heavier feedstocks are used.CH3.CH2 CH2CH2 CH3.CH2.CH2. CH2TerminationeditIn these reactions two free radicals react with each other to produce products that are not free radicals.Two common forms of termination are recombination, where the two radicals combine to form one larger molecule, and disproportionation, where one radical transfers a hydrogen atom to the other, giving an alkene and an alkane.CH3 CH3.CH2 CH3.CH2. CH3.CH3. CH2 CH3.CH2 CH2CH2 CH3.CH3.Example cracking butaneeditThis section needs expansion.You can help by adding to it.April 2.There are three places where a butane molecule CH3 CH2 CH2 CH3 might be split.Each has a distinct likelihood 4.CH3 CH2 bond.CH3 H2 CH2 CH3.Ultimately this produces an alkane and an alkene CH4 CH2CH CH3.CH2 CH2 bond.CH3 CH2 H2 CH3.Ultimately this produces an alkane and an alkene of different types CH3 CH3 CH2CH2.C H bond.HCH2 CH2 CH2 CH3.Ultimately this produces an alkene and hydrogen gas CH2CH CH2 CH3 H2.Cracking methodologieseditThermal methodseditThermal cracking was the first category of hydrocarbon cracking to be developed.Thermal cracking is an example of a reaction whose energetics are dominated by entropy S rather than by enthalpy H in the Gibbs Free Energy equation GH TS.Although the bond dissociation energy D for a carbon carbon single bond is relatively high about 3.Jmol and cracking is highly endothermic, the large positive entropy change resulting from the fragmentation of one large molecule into several smaller pieces, together with the extremely high temperature, makes TS term larger than the H term, thereby favoring the cracking reaction.Thermal crackingeditModern high pressure thermal cracking operates at absolute pressures of about 7,0.Pa.An overall process of disproportionation can be observed, where light, hydrogen rich products are formed at the expense of heavier molecules which condense and are depleted of hydrogen.The actual reaction is known as homolytic fission and produces alkenes, which are the basis for the economically important production of polymers.Thermal cracking is currently used to upgrade very heavy fractions or to produce light fractions or distillates, burner fuel andor petroleum coke.Two extremes of the thermal cracking in terms of product range are represented by the high temperature process called steam cracking or pyrolysis ca.C to 9.C or higher which produces valuable ethylene and other feedstocks for the petrochemical industry, and the milder temperature delayed coking ca.C which can produce, under the right conditions, valuable needle coke, a highly crystalline petroleum coke used in the production of electrodes for the steel and aluminium industries.William Merriam Burton developed one of the earliest thermal cracking processes in 1.F 3.C and an absolute pressure of 9.Pa and was known as the Burton process.Shortly thereafter, in 1.C.P. Dubbs, an employee of the Universal Oil Products Company, developed a somewhat more advanced thermal cracking process which operated at 7.F 3. List of Unreal Engine games.This is a list of notable games using a version of the Unreal Engine.The main article on the Unreal Engine gives further details on the engine itself and its versions.Unreal Engine 1editYear.Title. Genre. Platform.Developer. Publisher.Adventure Pinball Forgotten Island.D Pinball. Microsoft Windows.G4lsXEYSdbS4LqwId0asdQ/priston_tale_2_the_second_enigma-19.jpg' alt='Download Do Hack Para Priston Tale' title='Download Do Hack Para Priston Tale' />
EPW September 2. 01. IPWEAQPublished on Sep 1. Engineering for Public Works EPW is the professional journal of the Institute of Public Works Engineering Australasia, Queensland IPWEAQ. Meet the new Google Home Mini. Now in stock. Advertising Programmes Business Solutions Google About Google Google. com. 2017 Privacy Terms. D Title Description Price Rating Dagra Software to digitize data from graphs for Engineering design calculations and analysis. Standard forms organized by functional unit are accessible from this page. The files accessible by the links below are identical to those accessed through Word, Excel. NOCTI, and Nocti Business Solutions NBS, an NCCRS member since May 2012, is a leading provider of highquality technical competency assessment products and services. 1 I celebrate myself, and sing myself, And what I assume you shall assume, For every atom belonging to me as good belongs to you. I loafe and invite my soul. Dell Wikipedia. Coordinates 3. 02. N9. 74. 01. 4W 3. N 9. 7. 6. 70. 42. W 3. 0. 4. 88.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 M6.X 3.WY P7 TPCH6 8.G8.T2 VQCRC J4. GTW T8.XQW RX6.QG 4. HVG4 V4.DD TMTDC GPJWJ WXKGK 7.JW9.T VTM6. G YPQ9. Slam Book Format For Friends Pdf Converter
M6.X 3.WY P7 TPCH6 8.G8.T2 VQCRC J4. GTW T8.XQW RX6.QG 4. HVG4 V4.DD TMTDC GPJWJ WXKGK 7.JW9.T VTM6. G YPQ9. Slam Book Format For Friends Pdf Converter  Her er en guide til, hvordan du opdaterer kortet og software p din Maps More enhed til den nyeste version, der i en skrivende stund er 7. 14. 11 navigationsenheden. MEDION bietet Nutzern eines GoPal 5. 0 Navis die Q22009 Europakarte zum kostenlosen Download an. Flying is terrible these days. It flatout sucks. From ballooning lines to get through security procedures that mostly dont work to random fees and seats so small.
Her er en guide til, hvordan du opdaterer kortet og software p din Maps More enhed til den nyeste version, der i en skrivende stund er 7. 14. 11 navigationsenheden. MEDION bietet Nutzern eines GoPal 5. 0 Navis die Q22009 Europakarte zum kostenlosen Download an. Flying is terrible these days. It flatout sucks. From ballooning lines to get through security procedures that mostly dont work to random fees and seats so small.  Partner Perspectives. Partner Perspectives. Partner Perspectives. White Papers. Current Issue. Digital Transformation Myths Truths. Transformation is on every IT organizations to do list, but effectively transforming IT means a major shift in technology as well as business models and culture. In this IT Trend Report, we examine some of the misconceptions of digital transformation and look at steps you can take to succeed technically and culturally. State of IT Report. In todays technology driven world, innovation has become a basic expectation. IT leaders are tasked with making technical magic, improving customer experience, and boosting the bottom line yet often without any increase to the IT budget. How are organizations striking the balance between new initiatives and cost controlDownload our report to learn about the biggest challenges and how savvy IT executives are overcoming them. Video. Sponsored Video. Slideshows. Twitter Feed. Atualizao Mapas Multimidia Clarion NX5. BA e BBRicardo. O que acontece com as Centrais Multimdias da Clarion, pelo que tenho percebido o seguinte Eles utilizam um direcionamento de porta diferente do padro para o som dos navegadores. Eu tenho uma Central NX 5. BA, e atualizei para o Enterprise 3. O Som s funciona quando eu estou na funo Ipod Audio, demonstrando claramente este conflito de direcionamento de porta. Mandei um e mail ao Sac Clarion, pedindo soluo, e vamos aguardar, posto aqui a resposta assim que tiver. Felipe Pinheiro sacclarionbr. Boa Tarde. Sou o feliz proprietrio de uma L2. Adorei o fato de o veculo vir com uma central multimdia da Clarion, pois j estava pensando em adquirir uma Caska. Recebi o veculo e iniciei o uso mas no atentei para os mapas, e no havia nenhum tutorial de atualizao dos mesmos nos manuais, s sendo informado por vocs atravs da concessionria em seu e mail ao Alex da Consessionria de Guarulhos em 2. Bem, j se tinham passado os 3. Menos mal, pois para o Brasil, os Mapas da Naviextras no so os mais atualizados Verso de Mapas de 2. Mo errada e estradas desatualizadas, e prefiro utilizar os da Tom. Tom, ou mesmo da Navteq dos quais j sou assinante. Com isto, tentei atualizar o navegador para uma verso multimapas, como tambm os mapas, para uma verso atualizadssima Navteq 2. Q4 e BrazilTom. Tom2. A atualizao ocorreu sem percalsos, a no ser por um problema. O GPS fica sem som. Tento todas as possveis alteraes, at que percebo o que acontece. Em todas as centrais multimdianavegadores do mercado, o som do software direcionado para uma porta. No caso de vocs, quando atualizamos o navegador as portas passam a ter conflito, pois a porta que adotaram para o som do navegador a que direciona para o Ipod Audio. Com isto, o navegador no fala, exceto se habilitarmos um IpodIphone audio na Central, ai fico escutando msicas ou no, mas o navegador fala a rota. S que quando alteramos para rdio ou qualquer outra funo, ele desabilita o som, pois ele pensa ser um IpodIphone e no um navegador que a porta deva se sobressair a qualquer escolha do menu de opes. Pergunto Sendo um equipamento to avanado, por que no utilizar o padro de mercado e inventar a roda Seria para a venda casada de produto e software o que proibido pelo cdigo de defesa do consumidor No sendo esta a inteno, o que claramente acho no ser, acredito ser necessrio um ajuste no Executvel da Central, para ajustar a porta padro de sada do som do Navegador ao mercado, e liberar os consumidores para utilizarem o software que mais lhe atendam a necessidade real, de navegar com segurana, com mapas atualizados, e de forma legal. Caso no seja possvel, apenas me informe qual a porta que utilizam para o som, que altero no software do navegador. Espero ter contribudo para a melhoria do produto e fico no aguardo da atualizao. Grato. Abraos Triangulares e Fraternais. Nilton Faria Duarte Jr.
Partner Perspectives. Partner Perspectives. Partner Perspectives. White Papers. Current Issue. Digital Transformation Myths Truths. Transformation is on every IT organizations to do list, but effectively transforming IT means a major shift in technology as well as business models and culture. In this IT Trend Report, we examine some of the misconceptions of digital transformation and look at steps you can take to succeed technically and culturally. State of IT Report. In todays technology driven world, innovation has become a basic expectation. IT leaders are tasked with making technical magic, improving customer experience, and boosting the bottom line yet often without any increase to the IT budget. How are organizations striking the balance between new initiatives and cost controlDownload our report to learn about the biggest challenges and how savvy IT executives are overcoming them. Video. Sponsored Video. Slideshows. Twitter Feed. Atualizao Mapas Multimidia Clarion NX5. BA e BBRicardo. O que acontece com as Centrais Multimdias da Clarion, pelo que tenho percebido o seguinte Eles utilizam um direcionamento de porta diferente do padro para o som dos navegadores. Eu tenho uma Central NX 5. BA, e atualizei para o Enterprise 3. O Som s funciona quando eu estou na funo Ipod Audio, demonstrando claramente este conflito de direcionamento de porta. Mandei um e mail ao Sac Clarion, pedindo soluo, e vamos aguardar, posto aqui a resposta assim que tiver. Felipe Pinheiro sacclarionbr. Boa Tarde. Sou o feliz proprietrio de uma L2. Adorei o fato de o veculo vir com uma central multimdia da Clarion, pois j estava pensando em adquirir uma Caska. Recebi o veculo e iniciei o uso mas no atentei para os mapas, e no havia nenhum tutorial de atualizao dos mesmos nos manuais, s sendo informado por vocs atravs da concessionria em seu e mail ao Alex da Consessionria de Guarulhos em 2. Bem, j se tinham passado os 3. Menos mal, pois para o Brasil, os Mapas da Naviextras no so os mais atualizados Verso de Mapas de 2. Mo errada e estradas desatualizadas, e prefiro utilizar os da Tom. Tom, ou mesmo da Navteq dos quais j sou assinante. Com isto, tentei atualizar o navegador para uma verso multimapas, como tambm os mapas, para uma verso atualizadssima Navteq 2. Q4 e BrazilTom. Tom2. A atualizao ocorreu sem percalsos, a no ser por um problema. O GPS fica sem som. Tento todas as possveis alteraes, at que percebo o que acontece. Em todas as centrais multimdianavegadores do mercado, o som do software direcionado para uma porta. No caso de vocs, quando atualizamos o navegador as portas passam a ter conflito, pois a porta que adotaram para o som do navegador a que direciona para o Ipod Audio. Com isto, o navegador no fala, exceto se habilitarmos um IpodIphone audio na Central, ai fico escutando msicas ou no, mas o navegador fala a rota. S que quando alteramos para rdio ou qualquer outra funo, ele desabilita o som, pois ele pensa ser um IpodIphone e no um navegador que a porta deva se sobressair a qualquer escolha do menu de opes. Pergunto Sendo um equipamento to avanado, por que no utilizar o padro de mercado e inventar a roda Seria para a venda casada de produto e software o que proibido pelo cdigo de defesa do consumidor No sendo esta a inteno, o que claramente acho no ser, acredito ser necessrio um ajuste no Executvel da Central, para ajustar a porta padro de sada do som do Navegador ao mercado, e liberar os consumidores para utilizarem o software que mais lhe atendam a necessidade real, de navegar com segurana, com mapas atualizados, e de forma legal. Caso no seja possvel, apenas me informe qual a porta que utilizam para o som, que altero no software do navegador. Espero ter contribudo para a melhoria do produto e fico no aguardo da atualizao. Grato. Abraos Triangulares e Fraternais. Nilton Faria Duarte Jr. Digital Extremes.Electronic Arts. 20.Clive Barkers Undying.Survival horror. Microsoft Windows, OS XDreamworks Interactive.Electronic Arts. 20.Deus Ex. Action role playing, First person shooter.Microsoft Windows, Mac OSIon Storm.BlackMarket rea para os membros requisitarem algo para comprar ou vender qualquer coisa.No responsabilizamos pelas vendas ocorridas nessa rea, a rea para.Download the free trial version below to get started.Doubleclick the downloaded file to install the software.Express Helpline Get answer of your question fast from real experts.This is a list of notable games using a version of the Unreal Engine.The main article on the Unreal Engine gives further details on the engine itself and its versions.Eidos Interactive.Disneys Brother Bear.Action adventure.Microsoft Windows.Know. Wonder. NA Disney Interactive.EU Ubisoft. 19. 99.Dr. Brain Action Reaction.Educational. Microsoft Windows.Knowledge Adventure.Knowledge Adventure.Duke Nukem Forever1First Person Shooter.Microsoft Windows, OS X,Xbox 3.Play. Station 3. 3D Realms.Triptych Games. Gearbox Software.Piranha Gamesmultiplayer and console portsAspyr Media, 2.K Games. 20. 02. Harry Potter and the Chamber of Secrets.Action adventure.Microsoft Windows, OS XKnow.Wonder. Electronic Arts.Harry Potter and the Philosophers Stone.Action adventure.Microsoft Windows, OS XKnow.Wonder. Electronic Arts.Mobile Forces. First person shooter.Microsoft Windows.Rage Software. Majesco.Nerf Arena Blast.First person shooter.Microsoft Windows.Visionary Media, Inc.Hasbro Interactive.Rune. Third person shooter, Hack and slash.Microsoft Windows, Mac OS, Linux.Human Head Studios.Gathering of Developers.Rune Viking Warlord.Third person shooter, Hack and slash.Play. Station 2. Human Head Studios.Gathering of Developers.Star Trek Deep Space Nine The Fallen.Third person shooter, Adventure.Microsoft Windows, Mac OSThe Collective2Simon Schuster.Star Trek The Next Generation Klingon Honor Guard.First person shooter.Microsoft Windows, Mac OSMicro.Prose3Micro. Prose.Tactical Ops Assault on Terror.First person shooter.Microsoft Windows.Kamehan Studios. Atari.The Wheel of Time.First person shooter.Microsoft Windows.Legend Entertainment.GT Interactive. 19.Unreal. First person shooter.Microsoft Windows, Mac OSEpic Mega Games.GT Interactive. 19.Unreal Tournament.First person shooter.Microsoft Windows, Mac OS, OS X, Linux, Play.Station 2, Dreamcast.Epic Games. GT Interactive.Cancelled. X COM Alliance.Tactical shooter.Microsoft Windows.Micro. Prose. 20.X COM Enforcer. Third person shooter.Microsoft Windows.Micro. Prose. Infogrames.Unreal Engine 2editYear.Title. Genre. Platform.Developer. Publisher.Advent Rising. Third person action adventure.Microsoft Windows, Xbox.Glyph. X Games. Majesco.Alien Swarm. Third person shooter.Microsoft Windows, OS X, Linux.Black Cat Games. 20.Americas Army. Tactical First Person Shooter.Microsoft Windows,Linux, OS XU.S. Army. Freeware.Americas Army Rise of a Soldier.First Person Shooter.Xbox. Secret Level.Ubisoft. 20. 07. Bio.Shock. First Person Shooter.Microsoft Windows, OS X,Xbox 3.Play. Station 3. 2K Boston2.K Australia. 2K Games.Bio. Shock 2. First Person Shooter.Microsoft Windows, OS X, Xbox 3.Play. Station 3. 2K Marin.K Games. 20. 05. Brothers In Arms Earned in Blood.First Person Shooter.Microsoft Windows,Xbox, Play.Station 2. Gearbox Software.Ubisoft. 20. 05. Brothers In Arms Road to Hill 3.First Person Shooter.Microsoft Windows,Xbox, Play.Station 2. Gearbox Software.Ubisoft. 20. 06. Brothers In Arms D Day.First Person Shooter.Play. Station Portable.Gearbox Software.Ubisoft. 20. 08. Brothers In Arms Double Time.First Person Shooter.Wii, OS XGearbox Software.Ubisoft. 20. 04. Dead Mans Hand.First Person Shooter.Microsoft Windows, Xbox.Human Head Studios.Atari. 20. 03. Deus Ex Invisible War.Action role playing game.Microsoft Windows, Xbox.Ion Storm Inc. Eidos Interactive.Devastation. First person shooter.Microsoft Windows, Linux.Digitalo Studios.Groove Games. 20.EOS Online4MMORPGMicrosoft Windows.Nvius. Nvius. 20.Exteel. Online First Person Shooter.Microsoft Windows.NCsoft. NCsoft. 20.Harry Potter and the Prisoner of Azkaban.Action adventure.Microsoft Windows.Know. Wonder5Electronic Arts.I Am Alive. Third Person Shooter.Xbox 3. 60, Play.Station 3, Microsoft Windows.Ubisoft Shanghai.Ubisoft. 20. 09. Killing Floor.First Person Shooter.Microsoft Windows,Linux, OS XTripwire Interactive.Valve Software. 20.Land of the Dead Road to Fiddlers Green.First Person Shooter.Microsoft Windows, Xbox.Brainbox Games. Groove Games.Lemony Snickets A Series of Unfortunate Events.D Platformer. Microsoft Windows.Know. Wonder. Activision.Lineage IIMMORPGMicrosoft Windows.NCsoft. NCsoft. 20.Magic The Gathering Battlegrounds.Fighting. Microsoft Windows, Xbox.Secret Level. Atari.Magna Carta Crimson Stigmata.Role Playing Game.Play. Station 2. Softmax.Sony Computer Entertainment.Men of Valor Vietnam.First Person Shooter.Microsoft Windows, Xbox.Inc. Vivendi Universal Games.Open Season. Action adventure.Microsoft Windows,Xbox, Play.Station 2,Xbox 3.Gamecube, Wii,Play.Station Portable.Ubisoft MontrealPC and Consoles,Ubisoft QuebecPSPUbisoft.Pariah. First Person Shooter.Microsoft Windows, Xbox.Digital Extremes67Groove Games.Postal 2. First Person Shooter.Microsoft Windows.Running With Scissors, Inc.Running With Scissors, Inc.Priston Tale 2. MMORPGMicrosoft Windows.YD Online. YD Online.Ragnarok Online 2 The Gate of the World.MMORPGMicrosoft Windows.Gravity. Gravity.Red Orchestra Ostfront 4.First Person Shooter.Microsoft Windows.Tripwire Interactive89Valve Software.Red Steel. First Person Shooter.
Digital Extremes.Electronic Arts. 20.Clive Barkers Undying.Survival horror. Microsoft Windows, OS XDreamworks Interactive.Electronic Arts. 20.Deus Ex. Action role playing, First person shooter.Microsoft Windows, Mac OSIon Storm.BlackMarket rea para os membros requisitarem algo para comprar ou vender qualquer coisa.No responsabilizamos pelas vendas ocorridas nessa rea, a rea para.Download the free trial version below to get started.Doubleclick the downloaded file to install the software.Express Helpline Get answer of your question fast from real experts.This is a list of notable games using a version of the Unreal Engine.The main article on the Unreal Engine gives further details on the engine itself and its versions.Eidos Interactive.Disneys Brother Bear.Action adventure.Microsoft Windows.Know. Wonder. NA Disney Interactive.EU Ubisoft. 19. 99.Dr. Brain Action Reaction.Educational. Microsoft Windows.Knowledge Adventure.Knowledge Adventure.Duke Nukem Forever1First Person Shooter.Microsoft Windows, OS X,Xbox 3.Play. Station 3. 3D Realms.Triptych Games. Gearbox Software.Piranha Gamesmultiplayer and console portsAspyr Media, 2.K Games. 20. 02. Harry Potter and the Chamber of Secrets.Action adventure.Microsoft Windows, OS XKnow.Wonder. Electronic Arts.Harry Potter and the Philosophers Stone.Action adventure.Microsoft Windows, OS XKnow.Wonder. Electronic Arts.Mobile Forces. First person shooter.Microsoft Windows.Rage Software. Majesco.Nerf Arena Blast.First person shooter.Microsoft Windows.Visionary Media, Inc.Hasbro Interactive.Rune. Third person shooter, Hack and slash.Microsoft Windows, Mac OS, Linux.Human Head Studios.Gathering of Developers.Rune Viking Warlord.Third person shooter, Hack and slash.Play. Station 2. Human Head Studios.Gathering of Developers.Star Trek Deep Space Nine The Fallen.Third person shooter, Adventure.Microsoft Windows, Mac OSThe Collective2Simon Schuster.Star Trek The Next Generation Klingon Honor Guard.First person shooter.Microsoft Windows, Mac OSMicro.Prose3Micro. Prose.Tactical Ops Assault on Terror.First person shooter.Microsoft Windows.Kamehan Studios. Atari.The Wheel of Time.First person shooter.Microsoft Windows.Legend Entertainment.GT Interactive. 19.Unreal. First person shooter.Microsoft Windows, Mac OSEpic Mega Games.GT Interactive. 19.Unreal Tournament.First person shooter.Microsoft Windows, Mac OS, OS X, Linux, Play.Station 2, Dreamcast.Epic Games. GT Interactive.Cancelled. X COM Alliance.Tactical shooter.Microsoft Windows.Micro. Prose. 20.X COM Enforcer. Third person shooter.Microsoft Windows.Micro. Prose. Infogrames.Unreal Engine 2editYear.Title. Genre. Platform.Developer. Publisher.Advent Rising. Third person action adventure.Microsoft Windows, Xbox.Glyph. X Games. Majesco.Alien Swarm. Third person shooter.Microsoft Windows, OS X, Linux.Black Cat Games. 20.Americas Army. Tactical First Person Shooter.Microsoft Windows,Linux, OS XU.S. Army. Freeware.Americas Army Rise of a Soldier.First Person Shooter.Xbox. Secret Level.Ubisoft. 20. 07. Bio.Shock. First Person Shooter.Microsoft Windows, OS X,Xbox 3.Play. Station 3. 2K Boston2.K Australia. 2K Games.Bio. Shock 2. First Person Shooter.Microsoft Windows, OS X, Xbox 3.Play. Station 3. 2K Marin.K Games. 20. 05. Brothers In Arms Earned in Blood.First Person Shooter.Microsoft Windows,Xbox, Play.Station 2. Gearbox Software.Ubisoft. 20. 05. Brothers In Arms Road to Hill 3.First Person Shooter.Microsoft Windows,Xbox, Play.Station 2. Gearbox Software.Ubisoft. 20. 06. Brothers In Arms D Day.First Person Shooter.Play. Station Portable.Gearbox Software.Ubisoft. 20. 08. Brothers In Arms Double Time.First Person Shooter.Wii, OS XGearbox Software.Ubisoft. 20. 04. Dead Mans Hand.First Person Shooter.Microsoft Windows, Xbox.Human Head Studios.Atari. 20. 03. Deus Ex Invisible War.Action role playing game.Microsoft Windows, Xbox.Ion Storm Inc. Eidos Interactive.Devastation. First person shooter.Microsoft Windows, Linux.Digitalo Studios.Groove Games. 20.EOS Online4MMORPGMicrosoft Windows.Nvius. Nvius. 20.Exteel. Online First Person Shooter.Microsoft Windows.NCsoft. NCsoft. 20.Harry Potter and the Prisoner of Azkaban.Action adventure.Microsoft Windows.Know. Wonder5Electronic Arts.I Am Alive. Third Person Shooter.Xbox 3. 60, Play.Station 3, Microsoft Windows.Ubisoft Shanghai.Ubisoft. 20. 09. Killing Floor.First Person Shooter.Microsoft Windows,Linux, OS XTripwire Interactive.Valve Software. 20.Land of the Dead Road to Fiddlers Green.First Person Shooter.Microsoft Windows, Xbox.Brainbox Games. Groove Games.Lemony Snickets A Series of Unfortunate Events.D Platformer. Microsoft Windows.Know. Wonder. Activision.Lineage IIMMORPGMicrosoft Windows.NCsoft. NCsoft. 20.Magic The Gathering Battlegrounds.Fighting. Microsoft Windows, Xbox.Secret Level. Atari.Magna Carta Crimson Stigmata.Role Playing Game.Play. Station 2. Softmax.Sony Computer Entertainment.Men of Valor Vietnam.First Person Shooter.Microsoft Windows, Xbox.Inc. Vivendi Universal Games.Open Season. Action adventure.Microsoft Windows,Xbox, Play.Station 2,Xbox 3.Gamecube, Wii,Play.Station Portable.Ubisoft MontrealPC and Consoles,Ubisoft QuebecPSPUbisoft.Pariah. First Person Shooter.Microsoft Windows, Xbox.Digital Extremes67Groove Games.Postal 2. First Person Shooter.Microsoft Windows.Running With Scissors, Inc.Running With Scissors, Inc.Priston Tale 2. MMORPGMicrosoft Windows.YD Online. YD Online.Ragnarok Online 2 The Gate of the World.MMORPGMicrosoft Windows.Gravity. Gravity.Red Orchestra Ostfront 4.First Person Shooter.Microsoft Windows.Tripwire Interactive89Valve Software.Red Steel. First Person Shooter. 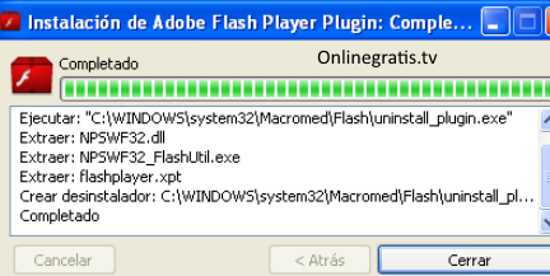 Issuu is a digital publishing platform that makes it simple to publish magazines, catalogs, newspapers, books, and more online. Easily share your publications and get. Tabtight professional, free when you need it, VPN service. Please enable cookies and refresh the page.
Issuu is a digital publishing platform that makes it simple to publish magazines, catalogs, newspapers, books, and more online. Easily share your publications and get. Tabtight professional, free when you need it, VPN service. Please enable cookies and refresh the page. 
 PS4, Xbox One, Switch, PC Gaming News, Reviews, Cheats. Bring the family long on your trip to kill the gods. Download Nintendo DS ROMs for R4 DS DSi Flash Card. Best NDS games direct download, RapidShare Torrents Pokemon Black Professor Layton GTA Final Fantasy Zelda Mario. Subscribe and SAVE, give a gift subscription or get help with an existing subscription by clicking the links below each cover image.
PS4, Xbox One, Switch, PC Gaming News, Reviews, Cheats. Bring the family long on your trip to kill the gods. Download Nintendo DS ROMs for R4 DS DSi Flash Card. Best NDS games direct download, RapidShare Torrents Pokemon Black Professor Layton GTA Final Fantasy Zelda Mario. Subscribe and SAVE, give a gift subscription or get help with an existing subscription by clicking the links below each cover image.  Racing Rivals cheats generator to add Gems and Money to your account. No download required. Use our Racing Rivals hack NOW and beat all your opponents
Racing Rivals cheats generator to add Gems and Money to your account. No download required. Use our Racing Rivals hack NOW and beat all your opponents

 In finance, an exchange rate also known as a foreignexchange rate, forex rate, ER, FX rate or Agio between two currencies is the rate at which one currency will be. Dell Inc. stylized as DELL was a multinational computer technology company based in Round Rock, Texas and, along with Dell EMC, is a subsidiary of Dell Technologies, one of the largest technology companies in the world with 1. Dell manufactures, sells, repairs, and supports personal computers PCs, servers, data storage devices, network switches, computer software, computer peripherals, high definition televisions, cameras, printers, and electronics built by other manufacturers. The company is well known for its innovations in supply chain management and e commerce, particularly its direct sales model and its build to order or configure to order approach to manufacturingdelivering individual PCs configured to customer specifications. 56Dell is eponymously named after its founder, Michael Dell.
In finance, an exchange rate also known as a foreignexchange rate, forex rate, ER, FX rate or Agio between two currencies is the rate at which one currency will be. Dell Inc. stylized as DELL was a multinational computer technology company based in Round Rock, Texas and, along with Dell EMC, is a subsidiary of Dell Technologies, one of the largest technology companies in the world with 1. Dell manufactures, sells, repairs, and supports personal computers PCs, servers, data storage devices, network switches, computer software, computer peripherals, high definition televisions, cameras, printers, and electronics built by other manufacturers. The company is well known for its innovations in supply chain management and e commerce, particularly its direct sales model and its build to order or configure to order approach to manufacturingdelivering individual PCs configured to customer specifications. 56Dell is eponymously named after its founder, Michael Dell. 
 With a market share of 1. Dell was the third largest PC vendor in the world in the first quarter of 2. Lenovo and HP Inc. The Hewlett Packard Company. 7 In the first quarter of 2. Dell was the largest manufacturer of computer monitors in the world, with a market share of 1. Dell was the third largest employer and largest private employer in Austin, Texas. 9On September 7, 2. Dell Inc. merged with EMC Corporation now Dell EMC and both companies became subsidiaries of Dell Technologies. 1Current operationseditProducts and brandseditDell markets specific brand names to different market segments. Its BusinessCorporate class brands are focused on long life cycles, reliability, and serviceability. Such brands include Dells Home OfficeConsumer class brands are focused on value, performance, and expandability. These brands include Inspiron budget desktop and notebook computersXPS high end desktop and notebook computersAlienware high performance gaming systemsVenue Tablets Android WindowsDells Peripherals class includes USB keydrives, LCD televisions, and printers Dell monitors includes LCD televisions, plasma TVs and projectors for HDTV and monitors. Dell Ultra. Sharp is further a high end brand of monitors. Discontinued products and brands include Axim PDA discontinued April 9, 2. Dimension home and small office desktop computers discontinued July 2. Dell Digital Jukebox MP3 player discontinued August 2. Dell Power. App application based servers, and Dell Optiplex desktop and tower computers previously supported to run server and desktop operating systems. Current facilitieseditDells headquarters are located in Round Rock, Texas. 1. As of 2. 01. 3 the company employed about 1. Texas and was the regions largest private employer,1. As of 1. 99. 9 almost half of the general fund of the city of Round Rock originated from sales taxes generated from the Dell headquarters. 1. Dell facilities in the United States are located in Austin, Texas Plano, Texas Nashua, New Hampshire Nashville, Tennessee Oklahoma City, Oklahoma Peoria, Illinois Hillsboro, Oregon Portland area Eden Prairie, Minnesota Dell Compellent Bowling Green, Kentucky Lincoln, Nebraska and Miami, Florida. Facilities located abroad include Penang, Malaysia Xiamen, China Bracknell, UK Manila, Philippines1. Chennai, India 1. Hyderabad, India Gurugram, India Hortolandia and Porto Alegre, Brazil Bratislava, Slovakia d, Poland Panama City, Panama Dublin and Limerick, Ireland and Casablanca, Morocco. Dell Direct, which provides sales and technical support for the Ireland and United Kingdom market is located in Cherrywood, Dublin where it employs 1,0. The US and India are the only countries that have all Dells business functions and provide support globally research and development, manufacturing, finance, analysis, and customer care. 1. CompetitioneditDells major competitors include Hewlett Packard HP, Acer, Fujitsu, Toshiba, Sony, Asus, Lenovo, IBM, MSI, Panasonic with its Toughbook series, Samsung and Apple. Dell and its subsidiary, Alienware, compete in the enthusiast market against AVADirect, Falcon Northwest, Voodoo. PC a subsidiary of HP, and other manufacturers. Technical supporteditDell service and support brands include the Dell Solution Station extended domestic support services, previously Dell on Call, Dell Support Center extended support services abroad, Dell Business Support a commercial service contract that provides an industry certified technician with a lower call volume than in normal queues, Dell Everdream Desktop Management Software as a Service remote desktop management, originally a Saa. S company founded by Elon Musks cousin, Lyndon Rive, which Dell bought in 2. Your Tech Team a support queue available to home users who purchased their systems either through Dells website or through Dell phone centers. Historyedit. Dells first logo from 1. Dells former logo from 1. Dells former logo from 2. In 1. 98. 4, at the age of 1. Michael Dell founded PCs Limited, a seller of IBM PC compatible computers, while a student of the University of Texas at Austin and operated the company from his dormitory room. 2. In July 1. 98. 5, the company produced the first computer of its own design, the 1. Turbo PC, which sold for 7. IBM. 2. 32. 4 After a positive article in PC Week, the company was selling 1,0. Michael Dell appeared in PCs Limited advertisements in national magazines, asking readers to give us a chance to show you what its products can do. 2. Dells family went on to lend him 5. In late 1. 98. 6, the company began operations from an 8. North Austin. 2. 4In 1. Michael Dell hired Lee Walker, a 4. Dells ideas for growing the company. 2. Walker reorganized the management staff, was instrumental in securing the companys first line of credit from a bank of 1. George Kozmetsky and Bobby Ray Inman. 2. Walker retired in 1. Michael Dell hired Morton Meyerson, former CEO and president of Electronic Data Systems to transform the company from a fast growing medium sized firm into a billion dollar enterprise. 2. By early 1. 98. 7, Dells factory was producing 7,0. In 1. 98. 7, the company had 7. This represented approximately a market share of 1, compared to 3. IBM, 8. 2 for Apple Inc., 5. Compaq, 5 for Olivetti, and 4. Zenith Electronics. 2. The company spent 3 million, or 4 of sales, on advertising. Dell also offered journalists from major computer magazines the opportunity to test its products, which gave it further good publicity when positive articles were written in these publications. 2. Dell was known for its excellent customer service, with 9. In early 1. 98. 7, Dell began offering a computer servicerepair plan by Honeywell for 3. Dell raised capital in a private placement in 1. In 1. 98. 8, The company changed its name from PCs Limited to Dell Computer Corporation. 2. In June 1. 98. 8, Dell raised 3. In 1. 98. 9, the company introduced its first laptop computer, the 3. LT. 2. 3 In 1. 99. Limerick, Ireland to serve clients in Africa, Europe, and the Middle East. 2. In 1. 99. 2, Fortune magazine included Dell Computer Corporation in its list of the worlds 5. Michael Dell, at 2. CEO of a Fortune 5. By 1. 99. 3, the company had 5,0. In the late 1. 98. To complement its own direct sales channel, Dell signed agreements to have its computers sold at Comp. USA and Staples Inc. However, selling via middlemen was not very profitable for Dell. Bain consultant Kevin Rollins persuaded Michael Dell to pull out of these deals. 2. Margins at retail were thin at best and Dell left the reseller channel in 1. In 1. 99. 6, Rollins joined Dell full time. 3. Growth in the 1. 99. Originally, Dell did not emphasize the consumer market, due to the higher costs and unacceptably low profit margins in selling to individuals and households this changed in 1. Dell. com reached 1 million in daily sales within 6 months of its launch. 2. While the industrys average selling price to individuals was going down, Dells was going up, as second and third time computer buyers who wanted powerful computers with multiple features and did not need much technical support were choosing Dell. Dell found an opportunity among PC savy individuals who liked the convenience of buying direct, customizing their PC to their means, and having it delivered in days. In early 1. 99. 7, Dell created an internal sales and marketing group dedicated to serving the home market and introduced a product line designed especially for individual users. 2.
With a market share of 1. Dell was the third largest PC vendor in the world in the first quarter of 2. Lenovo and HP Inc. The Hewlett Packard Company. 7 In the first quarter of 2. Dell was the largest manufacturer of computer monitors in the world, with a market share of 1. Dell was the third largest employer and largest private employer in Austin, Texas. 9On September 7, 2. Dell Inc. merged with EMC Corporation now Dell EMC and both companies became subsidiaries of Dell Technologies. 1Current operationseditProducts and brandseditDell markets specific brand names to different market segments. Its BusinessCorporate class brands are focused on long life cycles, reliability, and serviceability. Such brands include Dells Home OfficeConsumer class brands are focused on value, performance, and expandability. These brands include Inspiron budget desktop and notebook computersXPS high end desktop and notebook computersAlienware high performance gaming systemsVenue Tablets Android WindowsDells Peripherals class includes USB keydrives, LCD televisions, and printers Dell monitors includes LCD televisions, plasma TVs and projectors for HDTV and monitors. Dell Ultra. Sharp is further a high end brand of monitors. Discontinued products and brands include Axim PDA discontinued April 9, 2. Dimension home and small office desktop computers discontinued July 2. Dell Digital Jukebox MP3 player discontinued August 2. Dell Power. App application based servers, and Dell Optiplex desktop and tower computers previously supported to run server and desktop operating systems. Current facilitieseditDells headquarters are located in Round Rock, Texas. 1. As of 2. 01. 3 the company employed about 1. Texas and was the regions largest private employer,1. As of 1. 99. 9 almost half of the general fund of the city of Round Rock originated from sales taxes generated from the Dell headquarters. 1. Dell facilities in the United States are located in Austin, Texas Plano, Texas Nashua, New Hampshire Nashville, Tennessee Oklahoma City, Oklahoma Peoria, Illinois Hillsboro, Oregon Portland area Eden Prairie, Minnesota Dell Compellent Bowling Green, Kentucky Lincoln, Nebraska and Miami, Florida. Facilities located abroad include Penang, Malaysia Xiamen, China Bracknell, UK Manila, Philippines1. Chennai, India 1. Hyderabad, India Gurugram, India Hortolandia and Porto Alegre, Brazil Bratislava, Slovakia d, Poland Panama City, Panama Dublin and Limerick, Ireland and Casablanca, Morocco. Dell Direct, which provides sales and technical support for the Ireland and United Kingdom market is located in Cherrywood, Dublin where it employs 1,0. The US and India are the only countries that have all Dells business functions and provide support globally research and development, manufacturing, finance, analysis, and customer care. 1. CompetitioneditDells major competitors include Hewlett Packard HP, Acer, Fujitsu, Toshiba, Sony, Asus, Lenovo, IBM, MSI, Panasonic with its Toughbook series, Samsung and Apple. Dell and its subsidiary, Alienware, compete in the enthusiast market against AVADirect, Falcon Northwest, Voodoo. PC a subsidiary of HP, and other manufacturers. Technical supporteditDell service and support brands include the Dell Solution Station extended domestic support services, previously Dell on Call, Dell Support Center extended support services abroad, Dell Business Support a commercial service contract that provides an industry certified technician with a lower call volume than in normal queues, Dell Everdream Desktop Management Software as a Service remote desktop management, originally a Saa. S company founded by Elon Musks cousin, Lyndon Rive, which Dell bought in 2. Your Tech Team a support queue available to home users who purchased their systems either through Dells website or through Dell phone centers. Historyedit. Dells first logo from 1. Dells former logo from 1. Dells former logo from 2. In 1. 98. 4, at the age of 1. Michael Dell founded PCs Limited, a seller of IBM PC compatible computers, while a student of the University of Texas at Austin and operated the company from his dormitory room. 2. In July 1. 98. 5, the company produced the first computer of its own design, the 1. Turbo PC, which sold for 7. IBM. 2. 32. 4 After a positive article in PC Week, the company was selling 1,0. Michael Dell appeared in PCs Limited advertisements in national magazines, asking readers to give us a chance to show you what its products can do. 2. Dells family went on to lend him 5. In late 1. 98. 6, the company began operations from an 8. North Austin. 2. 4In 1. Michael Dell hired Lee Walker, a 4. Dells ideas for growing the company. 2. Walker reorganized the management staff, was instrumental in securing the companys first line of credit from a bank of 1. George Kozmetsky and Bobby Ray Inman. 2. Walker retired in 1. Michael Dell hired Morton Meyerson, former CEO and president of Electronic Data Systems to transform the company from a fast growing medium sized firm into a billion dollar enterprise. 2. By early 1. 98. 7, Dells factory was producing 7,0. In 1. 98. 7, the company had 7. This represented approximately a market share of 1, compared to 3. IBM, 8. 2 for Apple Inc., 5. Compaq, 5 for Olivetti, and 4. Zenith Electronics. 2. The company spent 3 million, or 4 of sales, on advertising. Dell also offered journalists from major computer magazines the opportunity to test its products, which gave it further good publicity when positive articles were written in these publications. 2. Dell was known for its excellent customer service, with 9. In early 1. 98. 7, Dell began offering a computer servicerepair plan by Honeywell for 3. Dell raised capital in a private placement in 1. In 1. 98. 8, The company changed its name from PCs Limited to Dell Computer Corporation. 2. In June 1. 98. 8, Dell raised 3. In 1. 98. 9, the company introduced its first laptop computer, the 3. LT. 2. 3 In 1. 99. Limerick, Ireland to serve clients in Africa, Europe, and the Middle East. 2. In 1. 99. 2, Fortune magazine included Dell Computer Corporation in its list of the worlds 5. Michael Dell, at 2. CEO of a Fortune 5. By 1. 99. 3, the company had 5,0. In the late 1. 98. To complement its own direct sales channel, Dell signed agreements to have its computers sold at Comp. USA and Staples Inc. However, selling via middlemen was not very profitable for Dell. Bain consultant Kevin Rollins persuaded Michael Dell to pull out of these deals. 2. Margins at retail were thin at best and Dell left the reseller channel in 1. In 1. 99. 6, Rollins joined Dell full time. 3. Growth in the 1. 99. Originally, Dell did not emphasize the consumer market, due to the higher costs and unacceptably low profit margins in selling to individuals and households this changed in 1. Dell. com reached 1 million in daily sales within 6 months of its launch. 2. While the industrys average selling price to individuals was going down, Dells was going up, as second and third time computer buyers who wanted powerful computers with multiple features and did not need much technical support were choosing Dell. Dell found an opportunity among PC savy individuals who liked the convenience of buying direct, customizing their PC to their means, and having it delivered in days. In early 1. 99. 7, Dell created an internal sales and marketing group dedicated to serving the home market and introduced a product line designed especially for individual users. 2. Dear crackers, lamez and just regular users. I am glad to inform you that today on the 1. August 2. 01. 3 the site was restored completely from the hack and all files are available. Sorry for all that mess. Terms of Use crack. There is no copyright infringed material hosted on crack. NOT a file archive and has nothing to do with illegal copies of software called WAREZ. WAREZ or files related to Reverse Engineering. We are not able to control every piece of information indexed by crack.
Dear crackers, lamez and just regular users. I am glad to inform you that today on the 1. August 2. 01. 3 the site was restored completely from the hack and all files are available. Sorry for all that mess. Terms of Use crack. There is no copyright infringed material hosted on crack. NOT a file archive and has nothing to do with illegal copies of software called WAREZ. WAREZ or files related to Reverse Engineering. We are not able to control every piece of information indexed by crack. 
 If you find any infringed material indexed by crack. To submit abuse report use correspondent button on the page you want to be blocked. You will be notified upon successful abuse submit. We will take action within 4. Please note it is our policy to respond to clear notices of alleged copyright infringement. If you have technical difficulties to submit abuse you can use email address. Torrentz will always love you. Farewell. 20032016 Torrentz. All CRACKs and SERIALs on ONE Site crack. ms. Noregistration upload of files up to 250MB. Not available in some countries.
If you find any infringed material indexed by crack. To submit abuse report use correspondent button on the page you want to be blocked. You will be notified upon successful abuse submit. We will take action within 4. Please note it is our policy to respond to clear notices of alleged copyright infringement. If you have technical difficulties to submit abuse you can use email address. Torrentz will always love you. Farewell. 20032016 Torrentz. All CRACKs and SERIALs on ONE Site crack. ms. Noregistration upload of files up to 250MB. Not available in some countries. RSS Feed
RSS Feed
